The Rise of Tubulysins: A New Frontier for High-Performance ADC Payloads
As the field of Antibody-Drug Conjugates (ADCs) evolves, the search for the next generation of high-performance payloads has turned toward tubulysins. Originally isolated from myxobacteria, these natural products are exceptionally potent antimitotics that disrupt microtubule dynamics, ultimately triggering apoptosis in cancer cells.
Tubulysins are rapidly gaining traction as a primary payload class due to their unique biological profile and the chemical flexibility they offer during conjugation.
Superior Potency and the Bystander Effect
What sets tubulysins apart from classical tubulin inhibitors—such as taxanes or vinca alkaloids—is their extraordinary cytotoxicity. These molecules frequently demonstrate picomolar to low-nanomolar potency, making them highly effective even at low concentrations.
Beyond sheer potency, tubulysins offer a strategic advantage in treating heterogeneous tumors. When integrated with cleavable linkers, their membrane-permeable metabolites can facilitate bystander killing. This allows the payload to exit the primary target cell and eliminate neighboring tumor cells that may not express the target antigen, thereby addressing one of the most significant challenges in oncology.
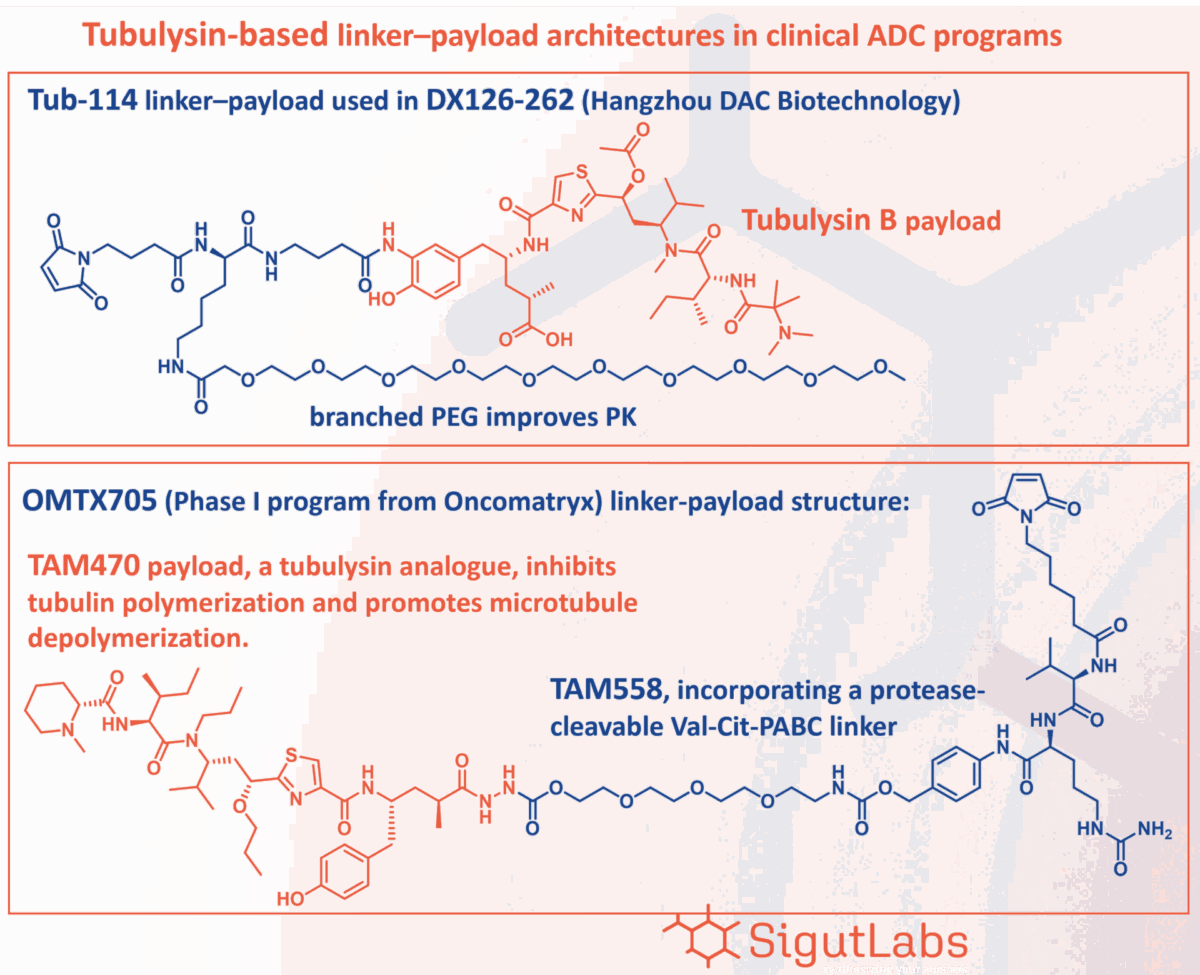
Clinical Benchmarks and Linker Innovation
Several clinical-stage programs underscore the potential of tubulysin-based ADCs and highlight the critical role of sophisticated linker chemistry:
OMTX705 (Oncomatryx): Currently in Phase I, this anti-FAP (Fibroblast Activation Protein) ADC utilizes a protease-cleavable Val-Cit-PABC/PABA linker motif. It carries a tubulysin/cytolysin payload (typically referenced as TAM470 or the related TAM558), demonstrating how established cleavage mechanisms can be paired with next-generation toxins.
DX126-262 (Hangzhou DAC Biotechnology): Reported in Phase II clinical trials, this candidate features a tubulysin B analogue known as Tub-114.
Engineering for Pharmacokinetic Success: The Tub-114 Model
The design of the Tub-114 linker-payload is particularly instructive for ADC developers. To optimize the molecule’s therapeutic profile, researchers introduced a branched PEG (polyethylene glycol) side chain. This specific modification serves two vital functions:
Enhanced Hydrophilicity: It improves the overall solubility of the conjugate.
Improved PK Behavior: By suppressing self-aggregation and reducing non-specific tissue uptake, the PEGylated linker ensures the ADC remains in circulation longer and reaches the target site more efficiently.
These examples reinforce the fact that payload chemistry and linker architecture are no longer independent variables; they must be co-engineered to define the success of the therapeutic.
Partnering with SigutLabs for Complex Synthesis
The transition to tubulysin payloads requires a high degree of chemical precision. At SigutLabs, we specialize in the most demanding aspects of ADC innovation: the design, synthesis, and optimization of custom linker-payload systems.
Our capabilities support partners throughout the development lifecycle, from the complex modification of cytotoxic payloads to the engineering of tailored conjugation handles and site-specific release mechanisms.
Are you evaluating tubulysins or other high-potency antimitotics for your ADC pipeline? Contact SigutLabs today to discuss how our synthesis expertise can help you optimize your linker-payload architectures.
OUR CASE STUDY
Scale-up to accelerate drug discovery
Our experience helped overcome development hurdles for potential cancer & mental health drugs.
Read moreEmpowering neuro research with pro-N6pA
Sigut Labs scaled up pro-N6pA production, simplifying AMPylation research & boosting accessibility.
Read moreADC development leaps with new linkers
Novel linker design expedited ADC advancement, leading to promising lead compounds faster.
Read moreLincomycin derivative scale-up
Over 30 g of the desired product with exceptional purity was obtained through our optimized procedure.
Read morePurifying 350 kg of vitamin K2 oil
Our innovative scale-up technology helped to reduce the client’s purification process from days to hours.
Read more
Custom synthesis
Providing for a custom synthesis of previously reported molecules using described synthetic procedures.

Contract research
Developing novel synthetic routes to provide undescribed compounds in organic, bioorganic, and medicinal chemistry.

Scale-Up
Helping you go from lab scales to an industrial scale by applying our cutting-edge instrumentation.

Our Experts


Partners & distributors











